Published on: June 21, 2023
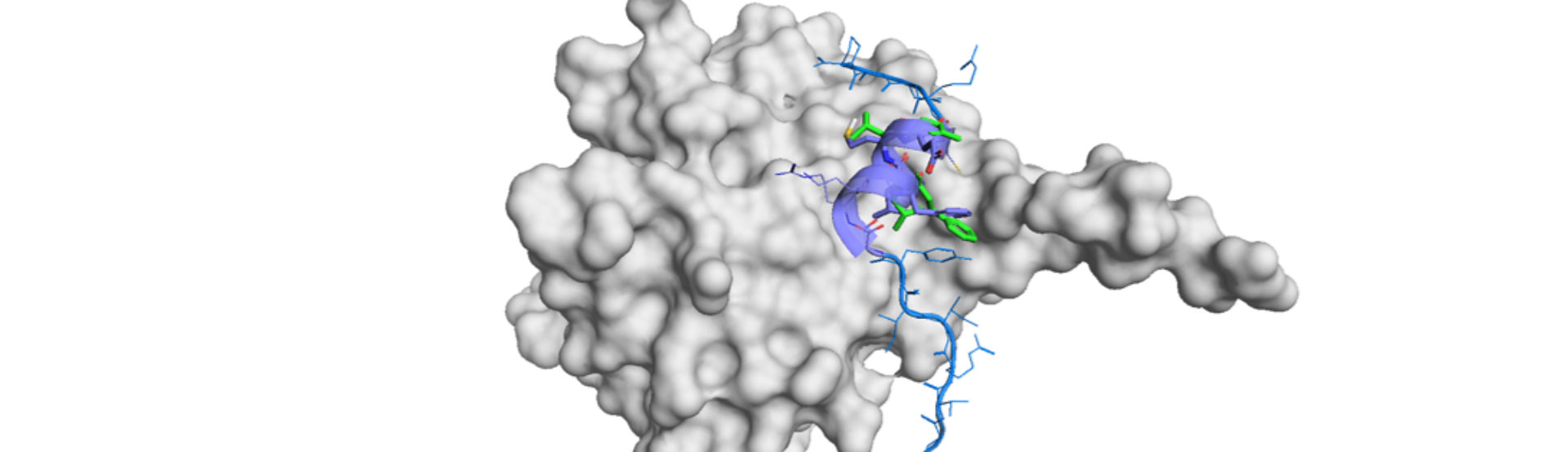
Conformational restriction has long been recognized in medicinal chemistry as a rational and effective way for structural design in both drug discovery and optimization stages. This strategy is crucial in PPIs in order for overcoming the energetical inferiority against the original, large-surface-based contact.
The exception might be the biomolecule drugs like antibodies which recognized the protein with a large surface area to achieve high affinity and selectivity. Antibody drugs are conceptually taking their role through the inhibition of the protein-biomolecule interaction including PPI. We thoroughly accept this irrefutable fact of the disadvantage of small molecules and reflect it in our peptidomimetic strategies and PepMetics® library,1)
Regardless of saying, peptidomimetics are intensive research field in 21st century.2) Peptidomimetics are advantageous over peptides in stability against proteolysis, tunability for cell permeation, potential readiness optimization for selectivity against off-targets and synthetic easiness if designed.
There are a couple of review articles on constrained peptides and conformational restriction strategies on peptides.3),4) In order to constrict the flexibility of linear peptides, macrocyclization and stapling are utilized so far and in some cases these strategies work well in not only the activity but also the pharmacokinetics and pharmacodynamics.
Cα alkylation, N-alkylation and Cβ alkylation are also utilized in combination with cyclization or stapling and there have been some successes. Stabilization by hydrogen bonds is another strategy to restrict the conformational flexibility.
Obviously, lots of strategies and approaches are available for “peptide-to-small molecule”, but there has been limited successes in drug discovery and development. Especially, orally available drugs are, at least so far, scarce when the project started from a peptide hit.
Our collaborator, Eisai inc. is currently conveying clinical trials with orally available compound with PepMetics® structure. PepMetics® molecules are significant in conformational stability with mild flexibility by molecule structure design.
There have been developed so many peptidomimetic scaffolds but most of them are flexible in nature. PepMetics® molecules basically have fixed structures as mimetics by tuning the sp3-enriched and conformationally restricted scaffolds.
This is a significance of our PepMetics®, we think. We are contiguously taking efforts on better scaffold development as well. Whenever you had an interest, please don’t hesitate to contact us for discussion.
1) https://doi.org/10.1021/acsomega.1c03967
2) https://doi.org/10.1039/d0cs00102c
3) https://doi.org/10.3390/molecules23040959
4) https://manu56.magtech.com.cn/progchem/EN/10.7536/PC150203#1