Published on: March 20, 2023
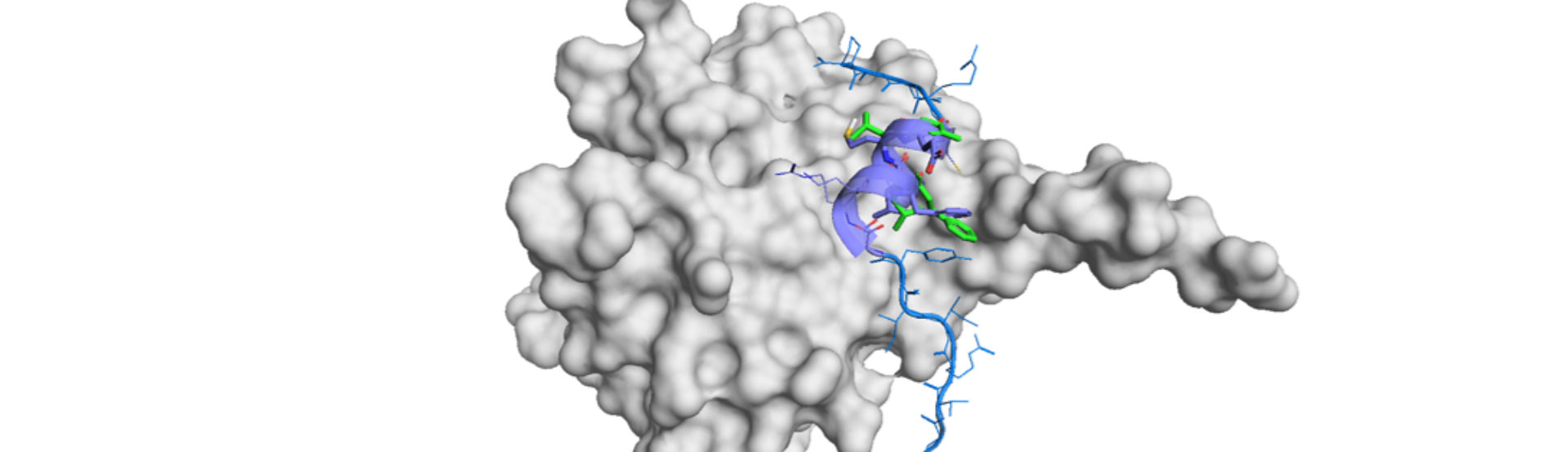
Visual analysis of proteins and molecule structures is a powerful methodology for better understanding and prediction of potential interactions and drug designs. In the field of PPIs, structural information of peptidomimetics is crucial for rapid drug discovery through designing a precise mimic of a protein or a peptide of interest. We developed two distinctive and complementary method for visual and quantitative analysis of peptidomimetics with mostly the same concept: peptide conformation distribution (PCD) plot and peptidomimetic analysis map (PMA).1) These versatile tools enable us comprehensive understanding of the characteristics of a peptidomimetic molecule and rational selection of the best scaffold for mimicking a peptide motif.
The PCD plot and the PMA map are generated by the examination of the side chain Cα-Cβ bonds of a peptide fragment motif and the corresponding (pseudo-Cα-Cβ) bonds of peptidomimetic molecules. The degree of the alignment of Cα-Cβ bonds represents the degree of mimicry of a peptidomimetic molecule since they define the chemical space of “side chain” of the peptide motif. It is a significant aspect because the conventional examination on φ and ψ angles of the main chain (i.e. Ramachandran plot2)) is not applicable for structural peptidomimetics, 3) which have original and unique scaffold for the replacement of peptide backbone.
The PCD plot is an alignment-free analysis of Cα and Cβ atom coordinates. It allows us visual inspection of the conformation distribution and the degree of chemical space coverage of peptidomimetic molecules as well as the characterization of an individual conformer. The concept of the PCD is simple: the projection of pseudo-Cα-Cβ coordinates of a peptidomimetic molecule to the real dataset of Cα-Cβ coordinate from PDB. The PCD plot is generated through principal component analysis with the support of USR (ultrafast shape recognition).4) It visually represents the nature of a peptidomimetic molecule based on the Cα-Cβ coordinate in an atomic level.
The PMA map, on the other hand, is focused analysis on a particular peptide fragment. This analysis is alignment-based because the result of the structural superposition of peptidomimetic molecules to the peptide of interest is visualized on the PMA map. This is a comparison method based on the Cα-Cβ position and direction (x-axis: the average of position difference (APD); y-axis: the average of vector difference (AVD)). The conformational variation is averaged for each molecule and compared against the peptide motif in this analysis. The PMA map is useful for comparison of different peptidomimetic scaffold because this map is independent of the mimetic patterns. A [i, i+1, i+2, i+3] mimetic molecule can readily be compared with [i, i+4, i+7] one on the same map, for example. This feature allows us rapid selection of the best mimetics of the target peptide by a simple visual inspection.
The PCD plot and the PMA map are not just a replacement of present structural analysis and visualization method. These analyses provide us quantitative and comprehensive evaluation of the characteristics and application opportunities of peptidomimetics based on the side chain Cα-Cβ parameters. Thus, the main chemical space of molecular recognition for PPIs is readily be understood by the use of the PCD plot and the PMA map. The combination of these complementary tools definitely accelerates PPI drug discovery. They are also boosting our technology, PepMetics®, by enhancing the scaffold development and expanding the chemical space as well. The PCD plot and the PMA map as well as PepMetics® would play an effective role in PPIs and peptidomimetics fields. If you have any questions on these technologies, please feel free to contact us to open up an opportunity for both of us.
1) https://doi.org/10.1021/acsomega.1c03967
2) https://doi.org/10.1107/S0907444902003359
3) https://doi.org/10.1002/anie.201412070
4) https://doi.org/10.1016/j.jmgm.2009.01.001