Published on: 1月 19, 2023
Novel technologies and strategies bring up beautiful sprouts of innovation. In our recent paper1, a two-step strategy for designing a small molecule was demonstrated by collaboration with GeneFrontier through the integration of respective technologies. This is a fantastic example of synergy creation, leveraging each strength to develop a new and practical strategy.
GeneFrontier has unique ribosome display system, PUREfrex®RD2, which has great advantage in screening of cyclic peptides from highly diversified library. While, we have PepMetics®, peptidomimetics for targeting PPIs and other biomolecule interactions.
The combination strategy of PUREfrex®RD and PepMetics® is as follows. PUREfrex®RD with huge library set of cyclic peptides allows swift screening and hit finding. This technology also enables SAR studies on their amino acid sequence of binder against the target as well. After obtaining the hit of a cyclic peptide, sequence mimetics, one of the outstanding characteristics of PepMetics®, is utilized to make it to a small molecule.
The question would be how to convert a cyclic peptide to a PepMetics® molecule. PepMetics® molecules are structural mimics of α-helix or β-turn of proteins. More precisely, they mimic the side chain Cα-Cβ bond of amino acids in α-helix or β-turn structures. Macrocyclic peptides generally form structural elements like a protein and the feature allows a simple, sequence-based transfer of a cyclic peptide sequence to PepMetics® molecules. A nominal number of molecules is necessary for the extraction of the essential sequence from a cyclic peptide. In order to mimic a cyclic peptide with 12 amino acids, a dozen or less are sufficient.
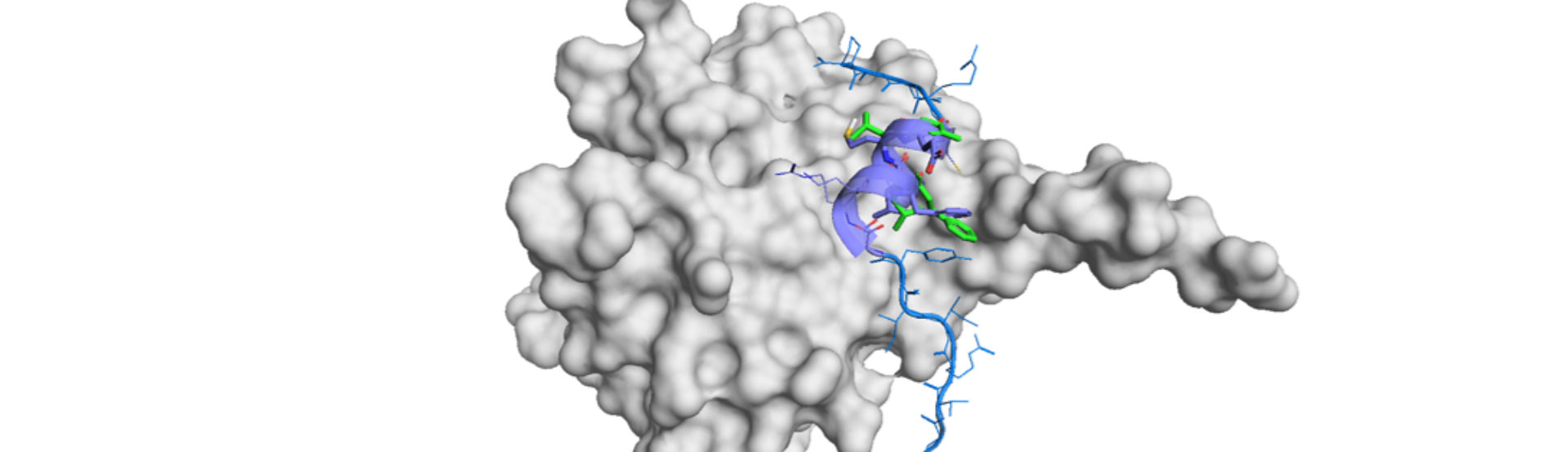
The effectiveness and usefulness of this rational and intuitive strategy is demonstrated by a rapid hit finding of CTLA-4 (cytotoxic T-lymphocyte-associated protein 4)-B7-1/B7-2 PPI inhibitor. The essence of this collaboration is as follows: optimization of PUREfrex®RD for screening of cyclic peptide, execution of screening campaign for CTLA-4 and hit finding, characterization of hit cyclic peptide with 12 residues and transformation by sequence mimetics. Identification of amino acid residues by amino acid substitutions of the cyclic peptides showed the overlap with the hit PepMetics® molecule. It should be emphasized that CTLA-4 is a much difficult target for discovering small molecule inhibitor even using fragment-based approach. This indicates that compounds with large molecular sizes are necessary regardless of ligand efficiency in order to find initial hit compounds against PPIs.
This cyclic peptide to small-molecule drug approach for a PPI inhibitor is realized by the combination of two technologies with high-compatibility. With a cyclic peptide binder in hand, sequence mimetics is applicable just by transferring the information of the amino acid to the small molecule design. We expect this collaborative approach with GeneFrontier is useful for small-molecule design targeting PPIs, even without structural information such as that associated with X-ray crystal structures.
This collaboration is giving us an opportunity to reconsider how to develop a technology. The combination of a few or more extinguished technology would produce a paradigm shift. Integration of reliable and versatile in vitro screening technology and small-molecule peptidomimetics technology is a fantastic strategy for PPI drug discovery, and potentially extended to a binder discovery against other biomolecules as well. We would continue to make a serious effort on development of useful technologies that enable more rapid and probable small-molecule candidates generation for drug development.