Published on: 2月 7, 2024
Macrocyclic drug has been intense research field, represented by macrocyclic peptides.1) It is promising if you see the presence of 67 FDA-approved macrocyclic drugs in 2023. but just over 90 compounds have been reported in approved drugs or those in the clinical stage.2)
One of the most examples is the Merck’s phase III case. Merck initiated phase III of MK-0616, an orally available macrocyclic peptide inhibitor of PCSK9 for atherosclerotic cardiovascular disease.3),4)
A macrocycle is still an emerging and underexploited chemical entity of intense interest. A macrocyclic compound has restricted conformation with a unique flexibility, controlled by the combination of atoms, bond types, the ring size as well as intramolecular interactions including hydrogen bonds and π-stackings.
However, the prediction of molecular properties including conformations, membrane permeability and stability is challenging and intense research has been conducted recently.5),6),7)
In addition, synthetic accessibility, especially on macrocyclization, is another issue of this molecular class due to the ring strain, transannular interactions, and conformational flexibility. In this sense, an interesting paper appeared in nature communications in 2023.8)
The authors developed a computational approach to design a macrocycle from an acyclic compound. The potential of their transformer architecture,9) Macformer, was demonstrated by design of macrocyclic JAK2 inhibitors by molecular docking.
Macformer is a deep generative model to design requisite linkers for macrocyclization. They collected 18,357 bioactive macrocyclic molecules with 12 or more in the ring size from ChEMBL database. Then they generated a pair dataset of acyclic and macrocyclic compound to use it for model training and evaluation.
The work frame of Macformer is friendly for medicinal chemists. Starting from a chemical structure of acyclic hit compound, you need to point out the position to attach a macrocycle linker. You may remove some substituents to pick out a requisite scaffold as well. After converting the molecular structure into SMILES, Macformer will generate the molecules reasonable linkers.
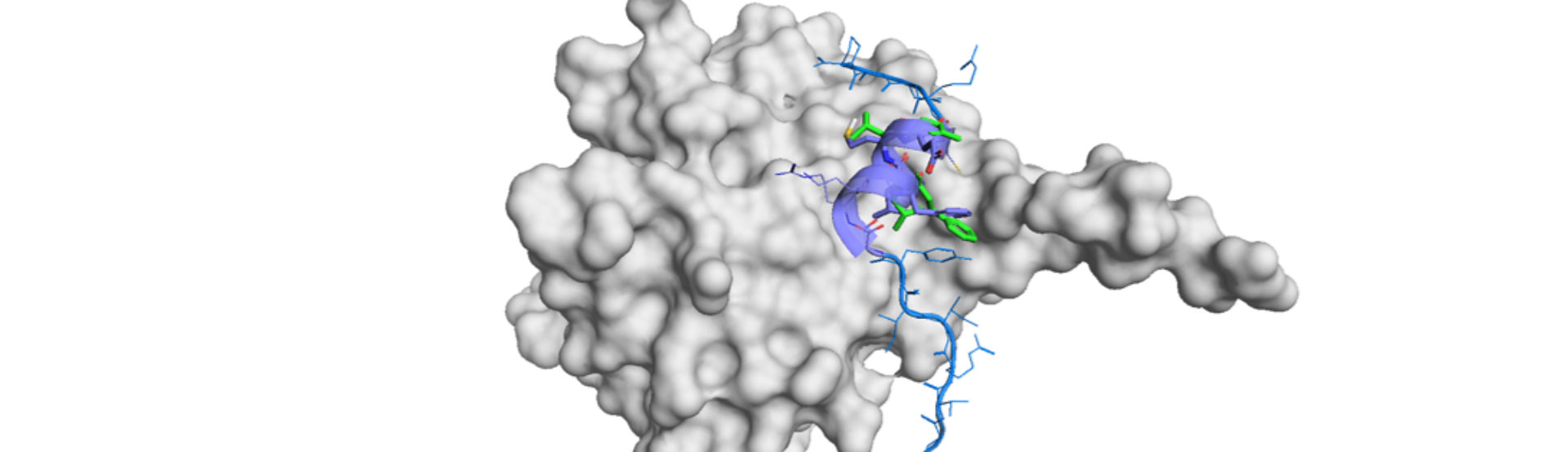
The authors took Fedratinib, a small molecule inhibitor of JAK2 for the model demonstration.10) Please see the literature for the detail of the process for the generation and filtration.
Basically, they generated 700 molecules and picked up 218 eligible macrocyclic compounds. Others were eliminated because of additional substituent groups were observed outside or inside the linker. Synthetic accessibility of macrocycles was evaluated on experience bases, and 3 compounds were picked and docked into JAK2. Visual inspection of docking models indicated very close binding interactions as the parent Fedrantinib.
Then they experimentally tested the three compounds 1-3 by enzyme assay, PK analyses and in vivo by a mouse IBD model. The enzyme inhibitory activity was not so significant (1: 0.070μM, 2: 0.364 μM and 3:0.006 μM against Fedratinib: 0.003 μM). Compound 3 had comparable activity and improved PK in terms of higher systemic exposure (AUCinf, 114.69 vs 50.19 h*ng/mL). In vivo activities of 3 was revealed to show a comparable therapeutic efficacy to Fedratinib at a lower dose in amelioration of the symptoms in DSS-induced murine colitis.
Macrocycle drug design by the use of Macformer still has problems because chemist needs to pick up reasonably synthesizable molecules with experience and intuitions. But it provides medicinal chemists possible designs of macrocycles and would be a useful tool for expanding chemical space of macrocycles.
https://doi.org/10.1038/nrd2590
https://doi.org/10.1021/acs.jmedchem.3c00134
https://www.merck.com/news/merck-initiates-phase-3-clinical-program-for-oral-pcsk9-inhibitor-candidate-mk-0616/
https://doi.org/10.1038/d41573-023-00152-3
https://doi.org/10.1007%2Fs11427-018-9342-2
https://doi.org/10.1038%2Fs41573-019-0050-3
https://doi.org/10.1038%2Fs42256-020-00236-4
https://doi.org/10.1038/s41467-023-40219-8
https://arxiv.org/abs/1706.03762
https://doi.org/10.1007%2Fs40265-019-01205-x